Baso4, also known as barium sulfate, is a white, odorless, and tasteless powder that has a variety of uses. It is a common ingredient in a variety of products, including paints, plastics, rubber, and cosmetics. In the medical field, it is often used as a contrast agent for x-ray and CT scans.
One of the main uses of baso4 is in the production of paints and coatings. It is a key ingredient in many types of paint because it is extremely opaque, meaning it is able to block out the color of whatever it is painted over. This makes it an excellent choice for use as a base coat or as a primer. It is also commonly used in the production of artist's paints because of its ability to block out the colors of the paints that are applied over it.
Baso4 is also used in the production of plastics and rubber. It is added to these materials to improve their durability and strength. It is also used as a filler in the production of plastic and rubber products to reduce the amount of raw materials needed, which can help to reduce costs.
In the cosmetics industry, baso4 is used as a thickening agent and a bulking agent. It is also used to improve the texture and appearance of cosmetics products, such as powders and foundations.
In the medical field, baso4 is commonly used as a contrast agent for x-ray and CT scans. It is administered orally or intravenously and helps to highlight specific areas of the body, making it easier for doctors to diagnose and treat certain conditions. It is also used in other medical procedures, such as angiography and gastrointestinal studies.
Overall, baso4 has a wide range of uses in a variety of industries. It is an important ingredient in many products and plays a vital role in the medical field as a contrast agent.
Barium Sulphate

It is the principal source of the element barium. Uses: Barium sulfate is widely used as a radio-opaque agent or X-ray contrast agent to diagnose gastrointestinal medical conditions. In nature, Barium sulfate is present in the Baryte mineral. It is a white crystalline solid that is odorless and insoluble in water. In addition to Barium sulfate, the clay particles will settle down, giving a clear colour of the indicator. Click here to learn more about Modified barium sulphate, high-quality barite powder procesed by special filtration and surface treatment technologies, has more excellent mechanical performances. Drink one-third of a bottle every fifteen minutes.
How is BaSO4 formed?

Its aqueous suspensions are neutral to litmus. Barium sulfate occurs as a fine, white, odorless, tasteless, bulky powder which is free from grittiness. It is removed by the body through a bowel movement. It was discovered by prominent English chemist and inventor, Sir Humphry Davy, 1st Baronet. What is the structural formula of barium sulfate? Can you drink water after drinking barium sulfate? As a result of a chemical reaction, atoms of chemical elements do not disappear anywhere and new ones do not appear, their number remains unchanged — this follows from the law of conservation of mass of substances. The chemical name of a compound is a way of identifying and describing the specific elements and their ratios that make up the compound.
Barium Sulphate (BaSo4), Barium Sulphate Manufacturers

The sulphur is attached to four oxygen atoms. The purified mineral is used as weighing mud during the drilling of oil wells because it increases the hydrostatic pressure and the density of the resultant liquid, effectively lowering the chances of a blowout. Salt can be formed from the acids and base reaction. The element barium is a silvery-white metal that is highly reactive and belongs to the alkaline earth metals group. Barium sulfate is likewise promptly expelled from the body, dissimilar to Thorotrast, which it replaced. It is managed orally, or by bowel purge, as a suspension of fine particles in a thick milk-like arrangement frequently with sweetening and seasoning agents added. Barium Sulfate Formula Barium sulfate is a significant inorganic chemical with several applications, including medical uses.
Barium sulfate
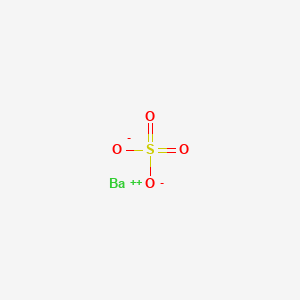
It is practically insoluble in water, solutions of acids and alkalies, and organic solvents. It is widely used in the production of oil and natural gas to get high-density drilling fluids by keeping the boreholes free of rock. It helps doctors study internal organs like the intestine, stomach, etc. In severe cases, a laxative may also be given to the patient. It is also insoluble in alcohols, and soluble in concentrated acids. Instructions for taking Oral Contrast Barium Sulfate Shake the contrast well before drinking. In the laboratory barium sulfate is generated by combining solutions of barium ions and sulfate salts.







