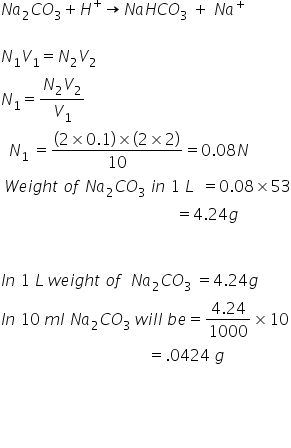Double indicator titration is a method used in chemistry to determine the concentration of a solution by measuring the volume of a known concentration of one solution required to react with a solution of unknown concentration. This method involves the use of two different indicators, one of which changes color at the beginning of the reaction and the other at the end.
The first step in a double indicator titration is to prepare the solutions that will be used in the reaction. The solution of unknown concentration is called the analyte, and the solution of known concentration is called the titrant. The two indicators are also prepared and added to the solutions.
The titration process begins by carefully measuring the volume of the titrant solution and adding it to the analyte solution in a graduated cylinder or burette. The solutions are mixed together, and the reaction between the two begins. As the reaction proceeds, the first indicator will change color, indicating that the reaction has reached the halfway point. The volume of titrant solution added up to this point is recorded.
The second indicator is then added to the solution, and the reaction continues until it is complete. As the reaction nears completion, the second indicator will change color, indicating that the endpoint has been reached. The volume of titrant solution added at this point is also recorded.
The concentration of the analyte solution can then be calculated using the volumes of titrant solution added at the halfway point and at the endpoint, as well as the known concentration of the titrant solution. This calculation allows for the determination of the concentration of the analyte solution with a high degree of accuracy.
Double indicator titration is a useful method for determining the concentration of solutions in a variety of applications, including in the analysis of chemical compounds and in the testing of water quality. It is a precise and reliable method that allows for the accurate measurement of solution concentrations in a variety of settings.
Double indicator titration is a laboratory technique that is used to determine the concentration of an unknown substance. It involves adding a known concentration of a second substance, called the titrant, to the unknown substance until the chemical reaction between the two is complete. This process is called titration.
In a double indicator titration, two different indicators are used to detect the endpoint of the reaction. The endpoint is the point at which the reaction is complete, and the concentration of the unknown substance can be calculated.
One indicator is used to detect the initial endpoint, while the second indicator is used to confirm the endpoint. This is important because it helps to ensure that the results of the titration are accurate and reliable.
The first indicator is chosen based on the chemical nature of the unknown substance and the titrant. It should have a distinct color change at the endpoint of the reaction. The second indicator is chosen based on the color change of the first indicator. It should have a different color change at the endpoint, but still be able to detect the endpoint of the reaction.
The double indicator titration process begins by preparing a solution of the unknown substance. A burette, a piece of laboratory equipment used to measure and dispense precise volumes of a liquid, is filled with the titrant. The titrant is then added to the unknown substance in small increments, while constantly stirring the mixture.
As the titrant is added, the color of the mixture will change due to the chemical reaction between the two substances. When the first indicator reaches its endpoint, the color of the mixture will change. The titrant is then added in small increments until the second indicator reaches its endpoint, at which point the color of the mixture will change again.
Once both indicators have reached their endpoint, the volume of titrant required to react with the unknown substance can be measured. This volume, along with the known concentration of the titrant, can be used to calculate the concentration of the unknown substance.
Double indicator titration is a valuable tool for determining the concentration of unknown substances. It is widely used in a variety of fields, including chemistry, biology, and pharmacology. It is especially useful when working with complex mixtures or when the endpoint of the reaction is difficult to detect.