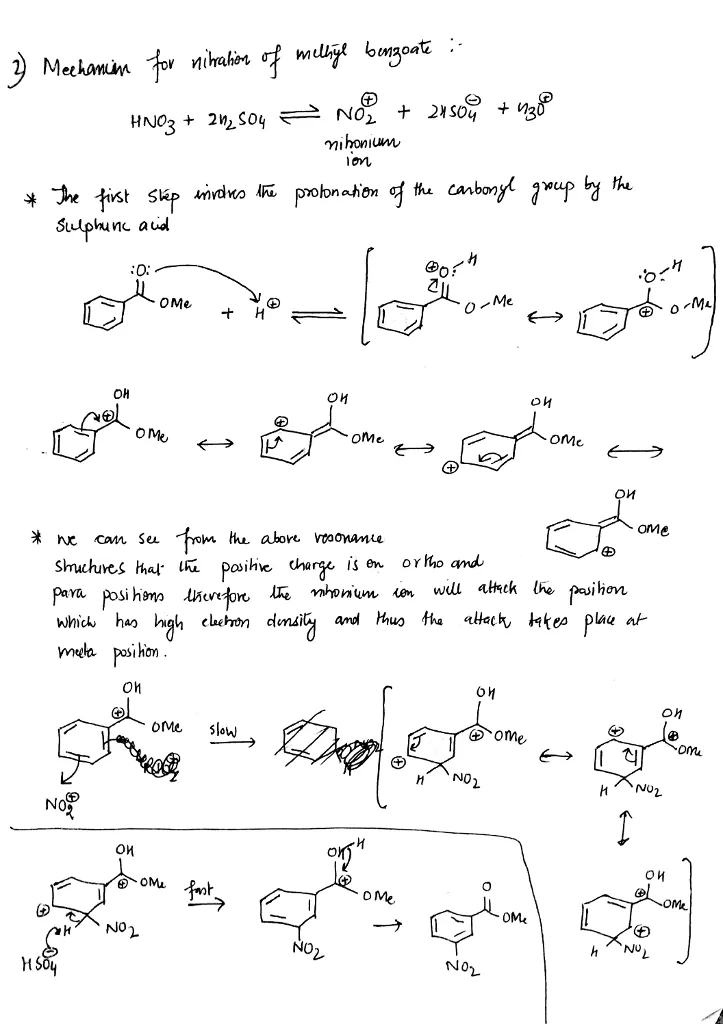
Nitration of methyl benzoate is a common laboratory experiment that is used to demonstrate the principles of electrophilic substitution reactions. In this reaction, methyl benzoate, a carbonyl compound, is converted into the corresponding nitro compound, nitro methyl benzoate, through the addition of a nitro group.
The reaction begins by mixing concentrated sulfuric acid with a small amount of nitrating agent, typically nitric acid. The sulfuric acid acts as a protonated catalyst, increasing the electrophilicity of the nitric acid and facilitating the attack of the nitro group on the double bond of the methyl benzoate.
After the reaction mixture has been stirred for a period of time, typically several hours, the product is isolated by adding the mixture to a large excess of cold water. The nitro methyl benzoate will crystallize out of solution, while the excess sulfuric acid and nitric acid will remain in the aqueous phase. The product can then be collected by filtration and dried.
One of the key features of the nitration of methyl benzoate is the regioselectivity of the reaction. The nitro group has a strong preference for attaching to the para position of the benzene ring, rather than the meta or ortho positions. This preference is due to the resonance stabilization of the intermediate species that is formed during the reaction.
The yield of the nitration of methyl benzoate can also be influenced by several factors, including the concentration of the reactants, the temperature of the reaction, and the presence of any solvents or catalysts. By carefully controlling these variables, it is possible to optimize the yield of the reaction and produce a high-quality product.
Overall, the nitration of methyl benzoate is a valuable laboratory experiment for demonstrating the principles of electrophilic substitution reactions and the factors that can influence the yield and regioselectivity of such reactions.
Private hospitals are healthcare facilities that are owned and operated by private organizations or individuals rather than by the government. These facilities can be for-profit or non-profit, and they offer a range of medical services to the public. Private hospitals are an important part of the healthcare system in many countries, as they provide alternative options to public hospitals and can often offer more specialized or advanced care.
One advantage of private hospitals is that they often have more resources and advanced technology at their disposal. This can allow them to offer a wider range of medical services, such as specialized surgeries or diagnostic tests, and to provide these services more quickly than public hospitals. Private hospitals may also be able to attract and retain top medical professionals, which can further improve the quality of care that they provide.
Private hospitals can also offer a more personalized and comfortable experience for patients. Many private hospitals have private rooms, which can be more comfortable and less stressful than shared rooms in public hospitals. Private hospitals may also offer amenities such as private bathrooms and meals prepared to the patient's specific dietary needs.
However, private hospitals can also have some disadvantages. One major concern is that they may be more expensive than public hospitals, which can be a barrier to access for some patients. Private hospitals may also prioritize profits over patient care, which can lead to unethical practices such as overcharging or unnecessary treatments.
Despite these concerns, private hospitals can play a valuable role in the healthcare system by providing specialized and advanced care and offering patients a more personalized experience. It is important for private hospitals to be regulated and held accountable to ensure that they are providing high-quality care and not prioritizing profits over patient welfare.
Nitration of methyl benzoate is a common laboratory experiment in undergraduate chemistry courses. It involves the reaction of methyl benzoate, a compound with a benzene ring and a methyl group attached, with nitric acid and sulfuric acid to form nitro methyl benzoate. This reaction is an example of an electrophilic substitution reaction, where the nitronium ion (NO2+) acts as the electrophile and the benzene ring of the methyl benzoate acts as the nucleophile.
The experiment begins by preparing a solution of nitric acid and sulfuric acid, known as "nitrating mixture." The nitrating mixture is typically prepared by carefully measuring out equal volumes of concentrated nitric acid and sulfuric acid and mixing them together. It is important to be cautious when handling these strong acids, as they can cause serious burns and are toxic if ingested.
Next, the methyl benzoate is added to the nitrating mixture and the reaction is allowed to proceed. The reaction typically occurs at room temperature, but it can be facilitated by heating the mixture. As the reaction proceeds, the nitronium ion attacks the benzene ring of the methyl benzoate, forming a nitro group and a carbocation intermediate. The intermediate is then stabilized by the methyl group, resulting in the formation of nitro methyl benzoate.
During the reaction, it is important to monitor the progress of the reaction using thin layer chromatography (TLC). TLC is a separation technique that allows for the identification and quantification of the various products formed during the reaction. By comparing the Rf values (a measure of the movement of a compound on the TLC plate) of the products to standards, the percentage yield of the nitro methyl benzoate can be determined.
Once the reaction is complete, the nitro methyl benzoate is typically purified by recrystallization. This involves dissolving the product in a solvent and allowing it to slowly crystallize out as the solution cools. The purified product is then collected, weighed, and analyzed using infrared spectroscopy to confirm the presence of the nitro group.
In conclusion, the nitration of methyl benzoate is a useful laboratory experiment that allows students to understand the mechanisms of electrophilic substitution reactions and the use of TLC for product analysis. It is important to be cautious when handling the strong acids used in the reaction and to follow proper safety protocols in the laboratory.