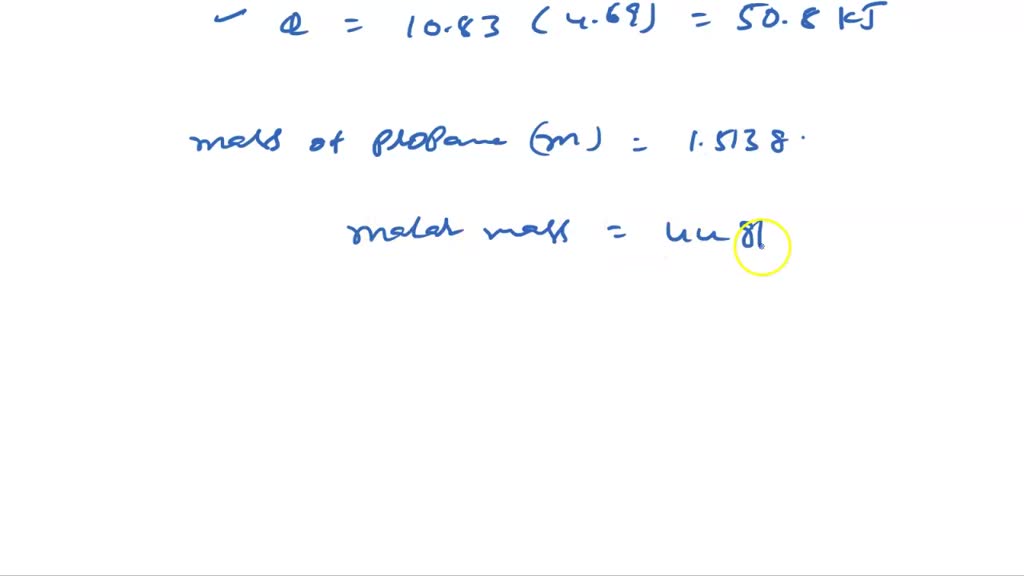Specific heat capacity of propanol. IPA 2023-01-01
Specific heat capacity of propanol
Rating:
4,9/10
1726
reviews
The specific heat capacity of a substance is a measure of the amount of energy required to raise the temperature of a given mass of that substance by a certain degree. It is an important property of a substance because it determines how quickly or slowly the substance will absorb or release heat when it is subjected to a temperature change. Propanol, also known as propyl alcohol or isopropyl alcohol, is a common solvent and is widely used in a variety of applications. In this essay, we will discuss the specific heat capacity of propanol and how it relates to the substance's properties and uses.
The specific heat capacity of propanol is 2.44 J/g·K. This means that it takes 2.44 joules of energy to raise the temperature of 1 gram of propanol by 1 degree Kelvin. Propanol has a relatively low specific heat capacity compared to other substances, which means that it absorbs and releases heat relatively quickly. This property makes it an effective solvent for dissolving a wide range of substances, as it can easily transfer heat to or from the dissolved material.
One of the primary uses of propanol is as a solvent for cleaning and degreasing surfaces. It is commonly used to remove dirt, oil, and other contaminants from a variety of surfaces, including metals, plastics, and glass. Propanol's low specific heat capacity makes it well suited for this purpose, as it can quickly dissolve and remove contaminants without significantly affecting the temperature of the surface being cleaned.
In addition to its use as a solvent, propanol is also used as a component of antifreeze and as a fuel additive. Its low specific heat capacity makes it an effective antifreeze, as it can absorb and release heat quickly to help prevent the formation of ice. As a fuel additive, propanol can improve the combustion characteristics of gasoline, making it more efficient and reducing emissions.
While propanol's low specific heat capacity is beneficial in many applications, it can also be a drawback in certain situations. For example, propanol's low heat capacity means that it can evaporate more quickly than other substances, which can make it less effective as a heat transfer medium in certain applications. It can also be more prone to overheating when used in high-temperature environments.
In conclusion, the specific heat capacity of propanol is an important property that affects its use and behavior. Its low specific heat capacity makes it an effective solvent and antifreeze, but it can also be a drawback in certain situations. Understanding the specific heat capacity of propanol and how it relates to its properties and uses is important for effectively utilizing this versatile substance.
What is the specific heat capacity of 2 propanol?

NIST Spectra 547 Program type: Complex; Column class: Semi-standard non-polar; Column diameter: 0. The energy content per volume unit of butanol is similar to the one of gasoline, and higher than the same for ethanol. Gupta V, Maken S, Kalra KC, Singh KC. Densities, speeds of sound, viscosities, and relative permittivities for 1-propanol + and 1-butanol + heptane at 298. Radon is a radioactive, colorless, odorless, tasteless noble gas.
Next
Table of specific heat capacities

The chemical symbol for Osmium is Os. Carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. The elemental metal is rarely found in nature, but once isolated artificially, the formation of an oxide layer passivation stabilizes the free metal somewhat against further oxidation. Fermium is a member of the actinide series. NIST Spectra 1072 Program type: Isothermal; Col umn class: Standard polar; Column length: 2 m; Column type: Packed; Start T: 75 C; CAS no: 71238; Active phase: Carbowax 20M; Carrier gas: N2; Substrate: Kieselgur 60-100 mesh ; Data type: Kovats RI; Authors: Goebel, K. For example, Paraffin has very large molecules and thus a high heat capacity per mole, but as a substance it does not have remarkable heat capacity in terms of volume, mass, or atom-mol which is just 1.
Next
Propane
Journal of Chemical Thermodynamics. Artigas H, Sanz ML, Mainar AM, Royo FM, Urieta JS. Uranium is weakly radioactive because all isotopes of uranium are unstable, with half-lives varying between 159,200 years and 4. From the combustion point of view, the production of carbon monoxide and volatile hydrocarbons from the combustion of alcohols is less than the one of gasoline. Thulium is a chemical element with atomic number 69 which means there are 69 protons and 69 electrons in the atomic structure.
Next
IPA

Romani L, Peleteiro J, Iglesias TP, Carballo E, Escudero R, Legido JL. Hwang S-C, Robinson Jr RL. Beryllium is a hard, grayish metal naturally found in mineral rocks, coal, soil, and volcanic dust. For example, 85% ethanol, E85, needs some modification of the internal combustion engines specifications, unlike butanols, which can work directly in present engines. Densities, sound speeds, excess volumes, and excess isentropic compressibilities of methyl acrylate + 1-propanol or 1-butanol + hydrocarbons n-hexane, n-heptane, cyclohexane, benzene, and toluene at 308. Like the other metals of the platinum group, ruthenium is inert to most other chemicals.
Next
Specific Heat Capacity of Chemical Elements

Vapour-liquid equilibrium of mixtures 1-propanol, or 1-butanol, + hydrocarbon With respect to the binary mixtures, Concerning temperature, most measurements were performed at low and moderate temperatures. Seven hydrocarbons linear, branched and cyclic alkanes, aromatics, and olefins have been selected as representative of present and future unleaded gasoline. Journal of Chemical Thermodynamics. The chemical symbol for Phosphorus is P. It is flammableand vapors are heavier than air. Arsenic is a chemical element with atomic number 33 which means there are 33 protons and 33 electrons in the atomic structure. Isothermal VLE and VE at 303.
Next
Acetone
The chemical symbol for Zirconium is Zr. What is the specific capacity of water? The chemical symbol for Aluminum is Al. Journal of Chemical Thermodynamics. Acetone is used as a solvent in paint and nail polish removers. Total vapour pressure measurements for binary mixtures of metanol, etanol, 1-propanol and 1-butanol with benzene, toluene and p-xylene at 313.
Next
Heat Capacity and Specific Heat Capacity

Journal of Chemical Thermodynamics. Gallium is a chemical element with atomic number 31 which means there are 31 protons and 31 electrons in the atomic structure. Commercially, thallium is produced as a byproduct from refining of heavy metal sulfide ores. Van Ness HC, Soczek CA, Peloquin GL, Machado RL. The chemical symbol for Palladium is Pd. Journal of Chemical and Engineering Data.
Next
Thermodynamic properties of normal propanol.
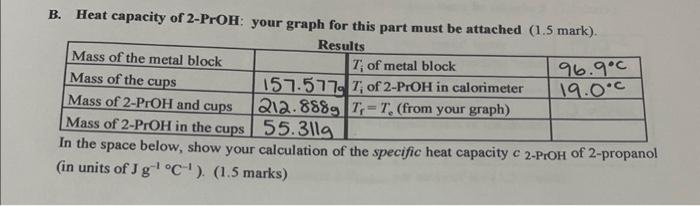
Limiting activity coefficients from an improved differential boiling point technique. Water is very resistant to changes in temperature, while metals generally are not. Isobaric vapor-liquid equilibrium of n-heptane-n-butanol system. A, 1160, 2007, 276-288. The chemical symbol for Barium is Ba.
Next