An essay outline is a helpful tool that helps writers organize their ideas and plan out the structure of their essay. It is a visual representation of the main points and arguments of the essay, arranged in a logical order. An essay outline can be as simple or as detailed as you need it to be, depending on the complexity of the essay and the requirements of your assignment.
There are several ways to format an essay outline, but one common method is the decimal outline, which uses a series of numbers and decimals to indicate the hierarchy of the main and supporting ideas. Here is a sample outline using the decimal format:
I. Introduction
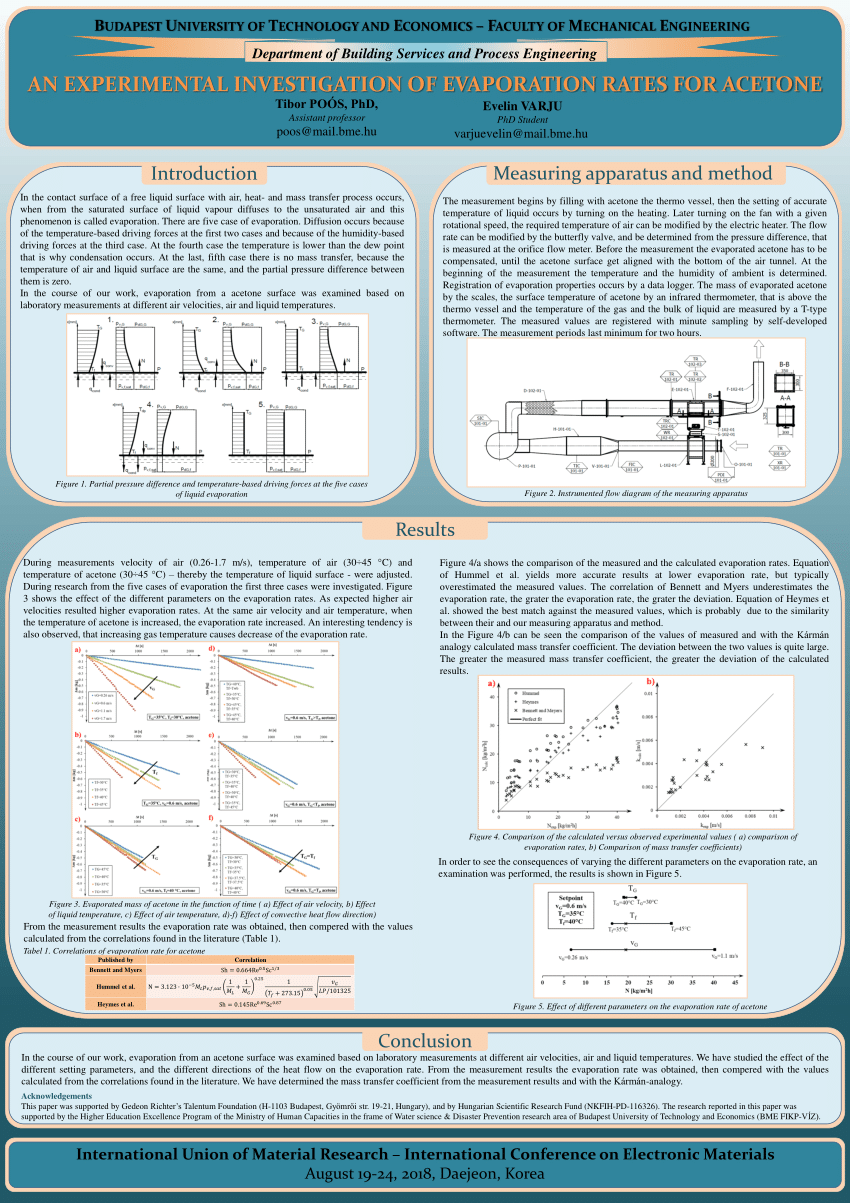
A. Hook
B. Thesis statement
C. Overview of main points
II. Body Paragraph 1
A. Main point 1
B. Supporting evidence
C. Explanation
III. Body Paragraph 2
A. Main point 2
B. Supporting evidence
C. Explanation
IV. Body Paragraph 3
A. Main point 3
B. Supporting evidence
C. Explanation
V. Conclusion
A. Restate thesis
B. Summary of main points
C. Final thoughts
As you can see, the outline begins with an introduction that includes a hook to grab the reader's attention, a thesis statement that states the main argument of the essay, and an overview of the main points that will be discussed. The body of the essay is then organized into paragraphs that each focus on a main point, with supporting evidence and explanation provided for each point. Finally, the conclusion restates the thesis and summarizes the main points, and offers final thoughts on the topic.
An essay outline is a useful tool for organizing your thoughts and keeping your essay on track. It helps you stay focused and ensures that all of your points are clearly and logically presented. By following a sample outline like the one above, you can easily create an outline for your own essay and start writing with confidence.
Why do liquids evaporate at different blog.sigma-systems.com
Requirement: Two Petri dishes of 5 cm. The computational thermal model has been shown to accurately track the transient thermal response of the test cells. Requirement: Three same size Petri dishes of diameter 10 cm, 10 ml. Chemistry Project Index Page no. In addition, molecules in motion have more energy than those at rest, and so the stronger the flow of air, the greater the evaporating power of the air molecules. In addition, it is illustrated that the maximum speed time-history of both gas and particle phases tend the same trend for small particle sizes. The results demonstrate that beside the values of particle cloud density, particle material density, and particle size, the action time is an important parameter in wave's suppression process, too.
Rate Of Evaporation Of Different Liquids [dvlryowxrp4z]

The rate of evaporation in an op en system is related to the vapour pressure found in a closed system. Notebook For Chemistry, Class XII Related Documents. If we change the volume of water for each beaker, the results we get will not be relevant, since we are seeing what affects the rate of evaporation, and the more the volume of water, the slower the rate of evaporation. The rate at which a liquid evaporates depends on its molecular structure. The cloud density, material density, and particle radius are the parameters considered in this analysis.
Rate of evaporation of different liquids?

In order to do so, the strength of intermolecular forces and the rate of evaporation for the volatile liquids must be determined first. In the US, the National Weather Service measures the actual rate of evaporation from a standardized "pan" open water surface outdoors, at various locations nationwide. As alcohol evaporates at a much faster rate compared with water due to its lower boiling temperature 82 compared to 100 degrees C , it is able to carry away more heat from the skin. It is recommended that the PTC-BC should be used for a spreading pool while the BR-BC should be used for a non-spreading pool. However, accidents during transportation and storage are a serious problem. Another approximation parameter was the evaporation velocity, defined as the evaporated volume of liquid per unit area of the liquid pool per unit time.