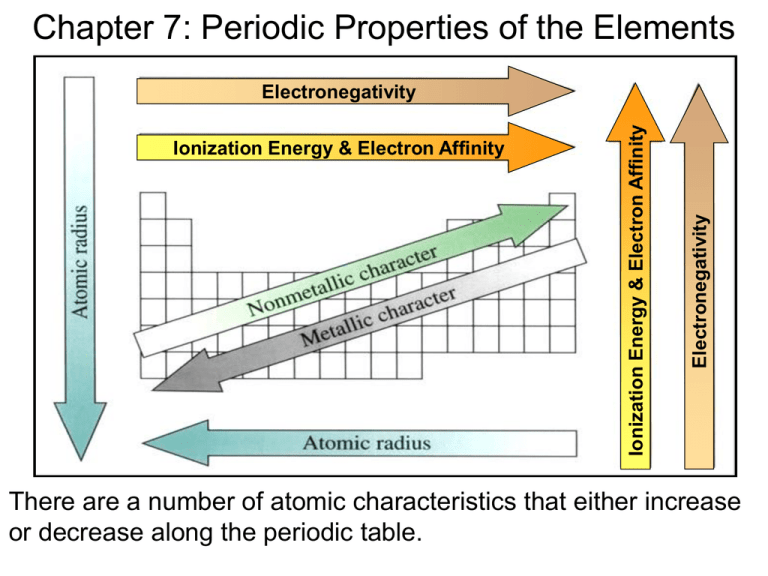
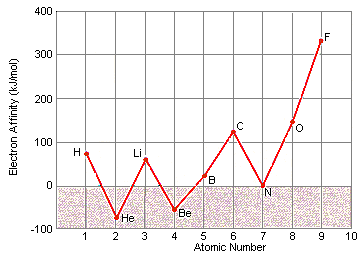
Sons and daughters are integral parts of any family, and their roles and responsibilities within the family dynamic can vary greatly depending on cultural and societal norms. In many societies, there are specific expectations and stereotypes placed on sons and daughters, which can shape their experiences and opportunities in life. In this essay, we will explore the ways in which sons and daughters are treated differently in various societies, and how these differences can impact their lives and relationships with their parents and siblings.
One of the most significant ways in which sons and daughters are often treated differently is in terms of their roles and responsibilities within the family. In many traditional societies, sons are often expected to take on more responsibilities and have more authority within the family, while daughters are expected to focus on domestic tasks and be more submissive to their male relatives. This can create a power dynamic within the family that can be difficult for daughters to navigate, and can also limit their opportunities and potential in life.
Additionally, sons and daughters may be treated differently in terms of their education and career opportunities. In some societies, daughters may be discouraged from pursuing higher education or certain careers, while sons may be encouraged to pursue these paths. This can create a gender-based divide in terms of what is expected of each child, and can have long-term consequences for their future prospects and success.
However, it is important to note that these gender-based expectations and stereotypes are not universal, and there are many societies where sons and daughters are treated more equally. In these societies, both boys and girls are encouraged to pursue their interests and dreams, regardless of gender. This can create a more equal and fair environment for both sons and daughters, and can help to break down the traditional gender roles and stereotypes that can limit the potential of both genders.
In conclusion, sons and daughters are often treated differently in various societies, and these differences can have a significant impact on their lives and opportunities. It is important to recognize and challenge these gender-based expectations and stereotypes, and to create a more equal and fair environment for both sons and daughters to thrive and reach their full potential.